| General Information of Drug (ID:
DR0106) |
| Drug Name |
Amphetamine
|
| Synonyms |
Actedron; Adipan; Allodene; Amfetamine; Anorexide; Anorexine; Benzebar; Benzedrine; Benzolone; Desoxynorephedrine; Elastonon; Fenopromin; Isoamyne; Isomyn; Mecodrin; Mydrial; Norephedrane; Novydrine; Oktedrin; Ortedrine; AMPHETAMINE; Percomon; Phenamine; Phenedrine; Profamina; Protioamphetamine; Simpatina; alpha-Methylbenzeneethaneamine; alpha-Methylphenethylamine; beta-Aminopropylbenzene; dl-Amphetamine; 1-Methyl-2-phenylethylamine; 1-Phenyl-2-aminopropane; 1-Phenyl-2-propylamine; 1-phenylpropan-2-amine; 300-62-9; Adderall; Finam
|
| Indication |
Attention deficit hyperactivity disorder
[ICD11: 6A05]
|
Approved
|
[1]
|
| Structure |
|
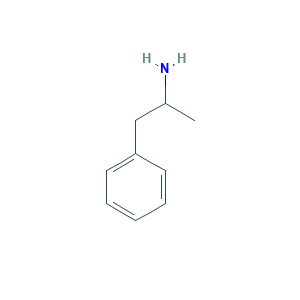 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
135.21 |
Topological Polar Surface Area |
26 |
| Heavy Atom Count |
10 |
Rotatable Bond Count |
2 |
| Hydrogen Bond Donor Count |
1 |
Hydrogen Bond Acceptor Count |
1 |
| Cross-matching ID |
- PubChem CID
- 3007
- PubChem SID
-
9717
; 87765
; 115861
; 608004
; 3249024
; 5340423
; 7978700
; 8150111
; 8151914
; 10517686
; 10524938
; 10529136
; 11336166
; 11361405
; 11462377
; 11533323
; 15297279
; 24882028
; 26746621
; 31073245
; 35804947
; 36519861
; 36651428
; 39909068
; 46506414
; 47216854
; 48110531
; 48415560
; 49855301
; 49855567
; 51091776
; 53789505
; 57321551
; 57651795
; 77830611
; 85209571
; 85756449
; 99302079
; 103164180
; 103810791
; 103918755
; 104302255
; 117525983
; 125680154
; 126677500
; 127272449
; 127272450
; 127272451
; 127272452
; 127272453
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D05BMG
- Formula
- C9H13N
- Canonical SMILES
- CC(CC1=CC=CC=C1)N
- InChI
- 1S/C9H13N/c1-8(10)7-9-5-3-2-4-6-9/h2-6,8H,7,10H2,1H3
- InChIKey
- KWTSXDURSIMDCE-UHFFFAOYSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


