| Synonyms |
Leroxacin; Lesacin; Levaquin; Levaquin (TN); Levofloxacin (INN); Levofloxacin [USAN:INN:JAN]; Levofloxacine; Levofloxacine [INN-French]; Levofloxacino; Levofloxacino [INN-Spanish]; Levofloxacinum; Levofloxacinum [INN-Latin]; Levokacin; Levoxacin; MP-376; Mosardal; Nofaxin; Ofloxacin S-(-)-form; Oftaquix; Oftaquix (TN); Quixin; Quixin (TN); R-Ofloxacin; Aeroquin; Cravit; Cravit (TN); Cravit Ophthalmic; D-Levofloxacin; DR 3354; DR-3355; DR-3355: L-isomer of ofloxacin; DR3355; Elequine; Floxacin; Floxel; Iquix (TN); L-Ofloxacin; RWJ 25213-097; RWJ-25213; Reskuin; S-(-)-Ofloxacin; Tavanic; Tavanic (TN); Volequin; (-)-(S)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido(1,2,3-de)-1,4-benzoxazine-6-carboxylic acid; (-)-(S)-9-fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyridol[1,2,3-de]-1,4-benzoxazine-6-carboxylic acid hemihydrate; (-)-(S)-9-fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7Hpyrido[1,2,3-de]-1,4-benzoxazine-6-carboxylic acid hemihydrate; (3S)-9-fluoro-3-methyl-10-(4-methylpiperazin-1-yl)-7-oxo-2,3-dihydro-7H-[1,4]oxazino[2,3,4-ij]quinoline-6-carboxylic acid; (R)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido(1,2,3-de)-1,4-benzoxazine-6-carboxylic acid; (R)-isomer; (S)-(-)-Ofloxacin; (S)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido(1,2,3-de)-1,4-benzoxazine-6-carboxylic acid; (S)-Ofloxacin; HR 355; HR-355; Iquix; LEVAQUIN IN DEXTROSE 5% IN PLASTIC CONTAINER; LFX; LVX; Levofloxacin tablet, suspension or intravenous; Levox
|
| Cross-matching ID |
- PubChem CID
- 149096
- PubChem SID
-
9862
; 598046
; 7979773
; 10250227
; 11364613
; 11367175
; 11369737
; 11372008
; 11374743
; 11377899
; 11485627
; 11489492
; 11490810
; 11492937
; 11495533
; 11528725
; 12014081
; 14876841
; 14901428
; 24857060
; 26612693
; 26680408
; 26719895
; 46225907
; 46386771
; 46505134
; 48185231
; 48334772
; 49665952
; 49681682
; 50064059
; 50123181
; 56314311
; 57346857
; 57648297
; 76034622
; 85261747
; 85789483
; 87558890
; 89736102
; 92124751
; 92307928
; 92308354
; 92309286
; 92710579
; 96024810
; 103165325
; 104052737
; 104179033
; 104253435
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D02RSN
- Formula
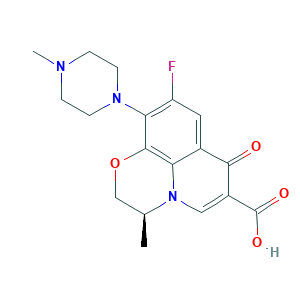
- C18H20FN3O4
- Canonical SMILES
- CC1COC2=C3N1C=C(C(=O)C3=CC(=C2N4CCN(CC4)C)F)C(=O)O
- InChI
- 1S/C18H20FN3O4/c1-10-9-26-17-14-11(16(23)12(18(24)25)8-22(10)14)7-13(19)15(17)21-5-3-20(2)4-6-21/h7-8,10H,3-6,9H2,1-2H3,(H,24,25)/t10-/m0/s1
- InChIKey
- GSDSWSVVBLHKDQ-JTQLQIEISA-N
|