| General Information of Drug (ID:
DR0420) |
| Drug Name |
Darunavir
|
| Synonyms |
Darunavir; Darunavirum; Darunavirum [INN-Latin]; MFCD09260006; NCGC00168773-01; TMC 114; TMC-114; TMC114; UIC-94017; UNII-YO603Y8113; YO603Y8113; (3R,3AS,6AR)-HEXAHYDROFURO[2,3-B]FURAN-3-YL(1S,2R)-3-[[(4-AMINOPHENYL)SULFONYL](ISOBUTYL)AMINO]-1-BENZYL-2-HYDROXYPROPYLCARBAMATE; (3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl ((2S,3R)-4-(4-amino-N-isobutylphenylsulfonamido)-3-hydroxy-1-phenylbutan-2-yl)carbamate; 206361-99-1; AIDS073035; CHEBI:367163; CHEMBL1323
|
| Indication |
Human immunodeficiency virus infection
[ICD11: 1C60]
|
Approved
|
[1]
|
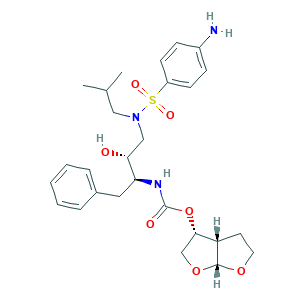
| Structure |
|
 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
547.7 |
Topological Polar Surface Area |
149 |
| Heavy Atom Count |
38 |
Rotatable Bond Count |
12 |
| Hydrogen Bond Donor Count |
3 |
Hydrogen Bond Acceptor Count |
9 |
| Cross-matching ID |
- PubChem CID
- 213039
- PubChem SID
-
628316
; 7884953
; 9371109
; 10322685
; 10322696
; 11108879
; 12015589
; 14719987
; 14719990
; 14720157
; 14788134
; 17397753
; 30417278
; 46394098
; 46394099
; 46394100
; 46394252
; 46394253
; 46394275
; 46394276
; 46506908
; 49661683
; 49743126
; 49743141
; 49743143
; 50070565
; 50071308
; 50074527
; 50113275
; 50333303
; 53777066
; 57399654
; 85098698
; 85177032
; 85177058
; 85756739
; 85756740
; 87351130
; 87557585
; 87557699
; 87557709
; 87557710
; 99226867
; 99226870
; 99226871
; 104223585
; 104234207
; 113453152
; 115001517
; 117695824
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D03IGH
- Formula
- C27H37N3O7S
- Canonical SMILES
- CC(C)CN(CC(C(CC1=CC=CC=C1)NC(=O)OC2COC3C2CCO3)O)S(=O)(=O)C4=CC=C(C=C4)N
- InChI
- 1S/C27H37N3O7S/c1-18(2)15-30(38(33,34)21-10-8-20(28)9-11-21)16-24(31)23(14-19-6-4-3-5-7-19)29-27(32)37-25-17-36-26-22(25)12-13-35-26/h3-11,18,22-26,31H,12-17,28H2,1-2H3,(H,29,32)/t22-,23-,24+,25-,26+/m0/s1
- InChIKey
- CJBJHOAVZSMMDJ-HEXNFIEUSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


