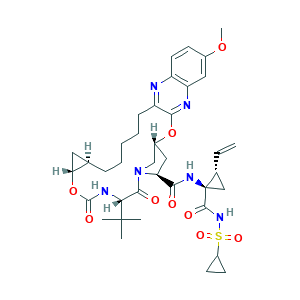
| Synonyms |
Grazoprevir; Grazoprevir [INN]; Grazoprevir anhydrous; MK 5172; MK-5172; MK5172; OBMNJSNZOWALQB-NCQNOWPTSA-N; SCHEMBL2175313; (1aR,5S,8S,10R,22aR)-5-tert-butyl-N-{(1R,2S)-1-[(cyclopropanesulfonyl)carbamoyl]-2-ethenylcyclopropyl}-14-methoxy-3,6-dioxo-1,1a,3,4,5,6,9,10,18,19,20,21,22,22a-tetradecahydro-8H-7,10-methanocyclopropa[18,19][1,10,3,6]dioxadiazacyclononadecino[11,12-b]quinoxaline-8-carboxamide; 1350514-68-9; 8YE81R1X1J; CHEBI:132975; CHEMBL2063090; DTXSID50159234; EX-A2253; UNII-8YE81R1X1J
|
| Cross-matching ID |
- PubChem CID
- 44603531
- ChEBI ID
-
- CAS Number
-
- Formula
- C38H50N6O9S
- Canonical SMILES
- CC(C)(C)C1C(=O)N2CC(CC2C(=O)NC3(CC3C=C)C(=O)NS(=O)(=O)C4CC4)OC5=NC6=C(C=CC(=C6)OC)N=C5CCCCCC7CC7OC(=O)N1
- InChI
- 1S/C38H50N6O9S/c1-6-22-19-38(22,35(47)43-54(49,50)25-13-14-25)42-32(45)29-18-24-20-44(29)34(46)31(37(2,3)4)41-36(48)53-30-16-21(30)10-8-7-9-11-27-33(52-24)40-28-17-23(51-5)12-15-26(28)39-27/h6,12,15,17,21-22,24-25,29-31H,1,7-11,13-14,16,18-20H2,2-5H3,(H,41,48)(H,42,45)(H,43,47)/t21-,22-,24-,29+,30-,31-,38-/m1/s1
- InChIKey
- OBMNJSNZOWALQB-NCQNOWPTSA-N
|