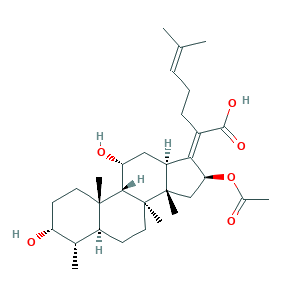
| Cross-matching ID |
- PubChem CID
- 3000226
- PubChem SID
-
8919
; 105658
; 596017
; 7979334
; 10036127
; 24702594
; 24894729
; 34666341
; 46392987
; 46505364
; 46530473
; 47206215
; 47736747
; 48416045
; 49829529
; 50959064
; 53787526
; 56314510
; 56320723
; 56320724
; 57410087
; 57654174
; 74382570
; 87895531
; 93576759
; 103511436
; 109693457
; 111610056
; 124766024
; 124801075
; 127344473
; 134338277
; 134988294
; 137004075
; 139685296
; 160965878
; 164814155
; 165235893
; 175266845
; 179230525
; 184811991
; 223715788
; 225067369
; 226413683
; 241109573
; 251916694
; 251917933
; 251970970
; 252350108
; 252402473
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D0X7XG
- Formula
- C31H48O6
- Canonical SMILES
- CC1C2CCC3(C(C2(CCC1O)C)C(CC4C3(CC(C4=C(CCC=C(C)C)C(=O)O)OC(=O)C)C)O)C
- InChI
- 1S/C31H48O6/c1-17(2)9-8-10-20(28(35)36)26-22-15-24(34)27-29(5)13-12-23(33)18(3)21(29)11-14-30(27,6)31(22,7)16-25(26)37-19(4)32/h9,18,21-25,27,33-34H,8,10-16H2,1-7H3,(H,35,36)/b26-20-/t18-,21-,22-,23+,24+,25-,27-,29-,30-,31-/m0/s1
- InChIKey
- IECPWNUMDGFDKC-MZJAQBGESA-N
|