Details of the Drug Metabolized by Drug-Metabolizing Enzyme (DME)
| General Information of Drug (ID: DR1362) | ||||||
|---|---|---|---|---|---|---|
| Drug Name |
HWA-285
|
|||||
| Synonyms |
Albert-285; Hextol; Karsivan; Propentofylina [INN-Spanish]; Propentofyllinum [INN-Latin]; Propentophylline; RBQOQRRFDPXAGN-UHFFFAOYSA-N; propentofylline; 1H-Purine-2,6-dione, 3,7-dihydro-3-methyl-1-(5-oxohexyl)-7-propyl-; 3,7-Dihydro-3-methyl-1-(5-oxohexyl)-7-propyl-1H-purine-2,6-dione; 3-Methyl-1-(5-oxohexyl)-7-propylxanthine; 3-methyl-1-(5-oxohexyl)-7-propyl-1H-purine-2,6(3H,7H)-dione; 55242-55-2; 5RTA398U4H; BRN 1156290; DSSTox_CID_25189; DSSTox_RID_80736; HOE-285; HWA 285; HWA-285; NCGC00015861-04; UNII-5RTA398U4H
|
|||||
| Indication | Cerebrovascular dementia [ICD11: 6D81] | Phase 1 | [1] | |||
| Structure |
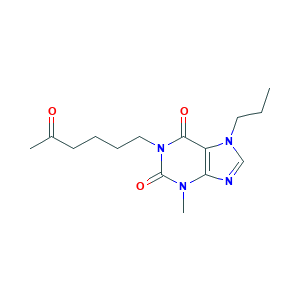 |
|||||
| 3D MOL | 2D MOL | |||||
| Pharmaceutical Properties | Molecular Weight | 306.36 | Topological Polar Surface Area | 75.5 | ||
| Heavy Atom Count | 22 | Rotatable Bond Count | 7 | |||
| Hydrogen Bond Donor Count | 0 | Hydrogen Bond Acceptor Count | 4 | |||
| Cross-matching ID |
|
|||||
| The Metabolic Roadmap of This Drug | |||||
|---|---|---|---|---|---|
| The Full List of Drug Metabolites (DM) of This Drug | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||
| The Full List of Metabolic Reaction (MR) of This Drug | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||
| Drug-Metabolizing Enzyme(s) (DME) Metabolizing This Drug | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.

