| Synonyms |
Ramosetron; LS-187182; NTHPAPBPFQJABD-LLVKDONJSA-N; Nor-YM 060; Ramosetron (INN); Ramosetron [INN]; SB19072; SC-92398; TL8000762; ZINC5116719; (-)-(R)-1-Methylindol-3-yl-4,5,6,7-tetrahydro-5-benzimidazolyl ketone; (1-methylindol-3-yl)-[(5R)-4,5,6,7-tetrahydro-3H-benzimidazol-5-yl]methanone; 132036-88-5; 7ZRO0SC54Y; 8235AH; A806353; AC1L3355; AJ-53160; AKOS015896003; BDBM50334454; CHEBI:135156; CHEMBL1643895; D08466; DB09290; DTXSID0043842; FT-0651831; GTPL2301; R-146; SCHEMBL16701; UNII-7ZRO0SC54Y
|
| Cross-matching ID |
- PubChem CID
- 108000
- PubChem SID
-
14824418
; 14848693
; 44436913
; 49830772
; 50070718
; 50112691
; 71879502
; 79053930
; 96025152
; 104380246
; 124974479
; 125084556
; 128751655
; 131297527
; 135141836
; 135650885
; 137446772
; 140430812
; 152035422
; 172659137
; 179149824
; 184546127
; 198983023
; 223562331
; 224977577
; 226406853
; 241031501
; 251912291
; 251916599
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D0RA9E
- Formula
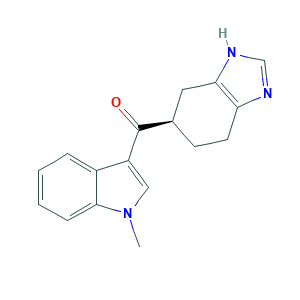
- C17H17N3O
- Canonical SMILES
- CN1C=C(C2=CC=CC=C21)C(=O)C3CCC4=C(C3)NC=N4
- InChI
- 1S/C17H17N3O/c1-20-9-13(12-4-2-3-5-16(12)20)17(21)11-6-7-14-15(8-11)19-10-18-14/h2-5,9-11H,6-8H2,1H3,(H,18,19)/t11-/m1/s1
- InChIKey
- NTHPAPBPFQJABD-LLVKDONJSA-N
|