Details of the Drug Metabolized by Drug-Metabolizing Enzyme (DME)
| General Information of Drug (ID: DR1946) | ||||||
|---|---|---|---|---|---|---|
| Drug Name |
Fenthion sulfoxide
|
|||||
| Synonyms |
FENTHION SULFOXIDE; FENTHION-SULFOXIDE; Fensulfoxide; Fenthionsulfoxide; Mesulfenfos; Mesulfenfos [ISO]; Mesulfenfos, (+)-; Mesulfenfos, (R)-; Mesulfenfos, (S)-; Mesulfenos; TM8HKU4HKN; 3761-41-9; BRN 2136026; O,O-Dimethyl O-((4-methylthio)-m-tolyl)phosphorothioate sulfoxide; O,O-Dimethyl O-(4-(methylsulfinyl)-m-tolyl) phosphorothioate; Phosphorothioic acid, O,O-dimethyl O-(3-methyl-4-(methylsulfinyl)phenyl) ester; Phosphorothioic acid, O,O-dimethyl O-(4-(methylsulfinyl)-m-tolyl) ester; UNII-J04Q566L5K; UNII-TM8HKU4HKN
|
|||||
| Indication | Discovery agent | Investigative | [1] | |||
| Structure |
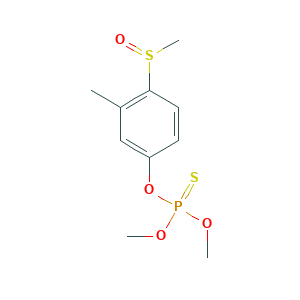 |
|||||
| 3D MOL | 2D MOL | |||||
| Pharmaceutical Properties | Molecular Weight | 294.3 | Topological Polar Surface Area | 96.1 | ||
| Heavy Atom Count | 17 | Rotatable Bond Count | 5 | |||
| Hydrogen Bond Donor Count | 0 | Hydrogen Bond Acceptor Count | 6 | |||
| Cross-matching ID | ||||||
| The Metabolic Roadmap of This Drug | |||||
|---|---|---|---|---|---|
| The Full List of Drug Metabolites (DM) of This Drug | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||
| The Full List of Metabolic Reaction (MR) of This Drug | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||
| Drug-Metabolizing Enzyme(s) (DME) Metabolizing This Drug | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||
| Experimental Enzyme Kinetic Data of This Drug | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||||||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.

