| General Information of Drug (ID:
DR2165) |
| Drug Name |
GW9662
|
| Synonyms |
Lopac-M-6191; Lopac0_000798; SCHEMBL420231; SR-01000075999; Spectrum5_001989; Tocris-1508; cid_644213; (2-chloro-5-nitrophenyl)-N-benzamide; 2-Chloro-5-nitro-N-4-phenylbenzamide; 2-Chloro-5-nitro-N-phenyl-benzamide; 2-Chloro-5-nitro-N-phenylbenzamide; 2-Chloro-5-nitrobenzanilide; 22978-25-2; AC1LD8S0; BSPBio_001021; CHEBI:79993; CHEMBL375270; DSSTox_CID_20723; DSSTox_GSID_40723; DSSTox_RID_79570; GTPL3442; GW 9662; GW-9662; GW9662; KBioGR_000361; KBioSS_000361; MLS001056751; SMR000326735; benzamide, 2-chloro-5-nitro-N-phenyl-
|
| Indication |
Breast cancer
[ICD11: 2C60]
|
Investigative
|
[1]
|
| Structure |
|
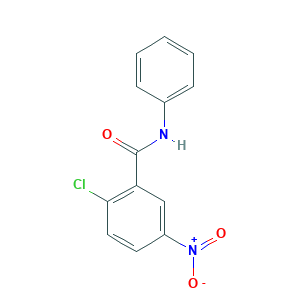 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
276.67 |
Topological Polar Surface Area |
74.9 |
| Heavy Atom Count |
19 |
Rotatable Bond Count |
2 |
| Hydrogen Bond Donor Count |
1 |
Hydrogen Bond Acceptor Count |
3 |
| Cross-matching ID |
- PubChem CID
- 644213
- PubChem SID
-
841875
; 877487
; 6873016
; 8707317
; 11111477
; 11114114
; 11121686
; 11122166
; 11362775
; 11365337
; 11367899
; 11370801
; 11370802
; 11373500
; 11376061
; 14824325
; 17396617
; 17405359
; 22395870
; 24278560
; 24883187
; 26732595
; 26752209
; 26752210
; 26752211
; 26758841
; 43765920
; 47719268
; 47793670
; 47942452
; 47942453
; 48392499
; 50104878
; 50104879
; 50104880
; 53777904
; 53790960
; 53800743
; 56355093
; 57408227
; 77530674
; 85202868
; 85231138
; 85787762
; 88369062
; 90341221
; 92304217
; 92310037
; 99300949
; 99302469
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D00ACX
- Formula
- C13H9ClN2O3
- Canonical SMILES
- C1=CC=C(C=C1)NC(=O)C2=C(C=CC(=C2)[N+](=O)[O-])Cl
- InChI
- 1S/C13H9ClN2O3/c14-12-7-6-10(16(18)19)8-11(12)13(17)15-9-4-2-1-3-5-9/h1-8H,(H,15,17)
- InChIKey
- DNTSIBUQMRRYIU-UHFFFAOYSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


