| General Information of Drug (ID:
DR5420) |
| Drug Name |
Pitolisant
|
| Synonyms |
Pitolisant; 362665-56-3; TIPROLISANT; BF2.649; UNII-4BC83L4PIY; 1-[3-[3-(4-chlorophenyl)propoxy]propyl]piperidine; Pitolisant (BF2.649); 1-(3-(3-(4-Chlorophenyl)propoxy)propyl)piperidine; 4BC83L4PIY; Piperidine, 1-[3-[3-(4-chlorophenyl)propoxy]propyl]-; CHEMBL462605; wakix; 1-[3-[3-(4-Chlorophenyl)propoxy]propyl]-piperidinehydrochloride; 1-{3-[3-(4-chlorophenyl)propoxy]propyl}piperidine; Tirolisant; Pitolisant (INN); tiprolisant [USAN]; Wakix (TN); Pitolisant [USAN:INN]; SCHEMBL117648; GTPL8924; CTK1B6404; HBS-101
|
| Indication |
Excessive daytime sleepiness
[ICD11: MG42]
|
Approved
|
[1]
|
|
Schizophrenia
[ICD11:
ICD11: 6A20]
|
Phase 3
|
[2]
|
| Structure |
|
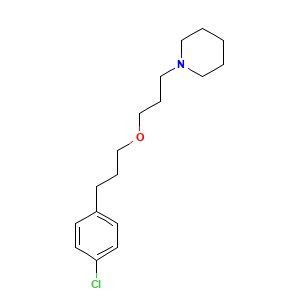 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
295.8 |
Topological Polar Surface Area |
12.5 |
| Heavy Atom Count |
20 |
Rotatable Bond Count |
8 |
| Hydrogen Bond Donor Count |
0 |
Hydrogen Bond Acceptor Count |
2 |
| Cross-matching ID |
- PubChem CID
- 9948102
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D01UUD
- Formula
- C17H26ClNO
- Canonical SMILES
- C1CCN(CC1)CCCOCCCC2=CC=C(C=C2)Cl
- InChI
- InChI=1S/C17H26ClNO/c18-17-9-7-16(8-10-17)6-4-14-20-15-5-13-19-11-2-1-3-12-19/h7-10H,1-6,11-15H2
- InChIKey
- NNACHAUCXXVJSP-UHFFFAOYSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


