| General Information of Drug (ID:
DR2287) |
| Drug Name |
Flucytosine
|
| Prodrug Info |
Flucytosine is the prodrug of Fluorouracil
|
| Synonyms |
Flucitosina; Flucitosina [DCIT]; Flucystine; Flucytosin; Flucytosine (JP15/USP/INN); Flucytosine [USAN:INN:BAN:JAN]; Flucytosinum; Flucytosinum [INN-Latin]; Flucytosone; Fluocytosine; Fluorcytosine; Fluorocytosine; Alcobon; Ancobon; Ancobon (TN); Ancotil; Ancotyl; Cytosine, 5-fluoro-(6CI,7CI,8CI); GL663142 & 5FC; LT00771985; Ro 2-9915; Ro 29915 E/265601; Ro-2-9915; 2(1H)-Pyrimidinone, 4-amino-5-fluoro-); 2-Hydroxy-4-amino-5-fluoropyrimidine; 4-Amino-5-fluoro-2(1H)-pyrimidinone; 4-Amino-5-fluoro-2-hydroxypyrimidine; 4-Amino-5-fluoro-2-hyroxypyrimidine; 4-Amino-5-fluoropyrimidin-2(1H)-one; 5-FC; 5-Flucytosine; 5-Fluorocystosine; 5-Fluorocytosin; 5-Fluorocytosine; 5-Fluorocytosine-6-3H; 5-Flurocytosine; 5-fluoro cytosine; 5987P; 6-Amino-2-oxo-5-fluoropyrimidine; 6-amino-5-fluoro-1H-pyrimidin-2-one; 9074P; F0321; GL663142 & 4-Amino-5-fluoropyrimidin-2(1H)-one
|
| Indication |
Cryptococcosis
[ICD11: 1F27]
|
Approved
|
[1]
|
| Structure |
|
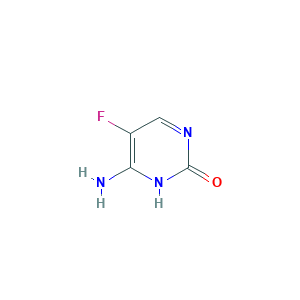 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
129.09 |
Topological Polar Surface Area |
67.5 |
| Heavy Atom Count |
9 |
Rotatable Bond Count |
0 |
| Hydrogen Bond Donor Count |
2 |
Hydrogen Bond Acceptor Count |
3 |
| Cross-matching ID |
- PubChem CID
- 3366
- PubChem SID
-
405592
; 602763
; 841044
; 1434447
; 3140368
; 3218074
; 5270497
; 5329900
; 5431724
; 6041883
; 7847389
; 7979233
; 8139861
; 11112505
; 11404488
; 11466962
; 11468082
; 11486773
; 11564865
; 14747633
; 15194483
; 24712291
; 24870520
; 24894919
; 24894928
; 26719629
; 29222501
; 39910157
; 46386766
; 46504735
; 47662407
; 47736597
; 48110551
; 48110552
; 48416018
; 49699216
; 49831643
; 49960195
; 51074762
; 53653662
; 56424024
; 57321755
; 57930982
; 81041221
; 83591697
; 85086121
; 87570095
; 87633378
; 88817477
; 92125834
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D0S5WG
- Formula
- C4H4FN3O
- Canonical SMILES
- C1=NC(=O)NC(=C1F)N
- InChI
- 1S/C4H4FN3O/c5-2-1-7-4(9)8-3(2)6/h1H,(H3,6,7,8,9)
- InChIKey
- XRECTZIEBJDKEO-UHFFFAOYSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


