| General Information of Drug (ID:
DR1620) |
| Drug Name |
Torasemide
|
| Synonyms |
Torasemida; Torasemida [INN-Spanish]; Torasemide; Torasemide, Anhydrous; Torasemidum; Torasemidum [INN-Latin]; Torsemide (Demadex); Demadex; JDL 464; JDL-464; Luprac; TORSEMIDE; W31X2H97FB; 1-Isopropyl-3-((4-m-toluidino-3-pyridyl)sulfonyl)urea; 56211-40-6; AC 4464; AC-4464; AC4464; BM 02015; BM-02015; BRN 0498515; CCRIS 6736; CHEBI:9637; MLS001165687; N-(((1-Methylethyl)amino)carbonyl)-4-((3-methylphenyl)amino)-3-pyridinesulfonamide; N-(Isopropylcarbamoyl)-4-(m-tolylamino)pyridine-3-sulfonamide; Torem; UNII-W31X2H97FB
|
| Indication |
Congestive heart failure
[ICD11: BD10]
|
Approved
|
[1]
|
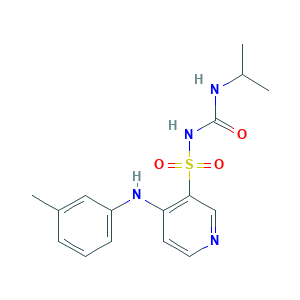
| Structure |
|
 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
348.4 |
Topological Polar Surface Area |
109 |
| Heavy Atom Count |
24 |
Rotatable Bond Count |
5 |
| Hydrogen Bond Donor Count |
3 |
Hydrogen Bond Acceptor Count |
5 |
| Cross-matching ID |
- PubChem CID
- 41781
- PubChem SID
-
7585786
; 7847448
; 8177279
; 11112793
; 11364670
; 11367232
; 11369794
; 11373102
; 11373959
; 11377957
; 11467058
; 11468178
; 11484799
; 11486816
; 11488958
; 11491647
; 11492105
; 11495590
; 11528723
; 12012656
; 14900728
; 25819958
; 26612831
; 26680347
; 26719820
; 26748988
; 26748989
; 34707389
; 46386585
; 46504760
; 47291390
; 47515595
; 47515596
; 47736767
; 48259511
; 48334790
; 48416649
; 49665992
; 49666477
; 49681707
; 49699258
; 49833537
; 50107501
; 53789088
; 56313713
; 57312605
; 80124867
; 81040892
; 85788916
; 87560438
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D0J9XZ
- Formula
- C16H20N4O3S
- Canonical SMILES
- CC1=CC(=CC=C1)NC2=C(C=NC=C2)S(=O)(=O)NC(=O)NC(C)C
- InChI
- 1S/C16H20N4O3S/c1-11(2)18-16(21)20-24(22,23)15-10-17-8-7-14(15)19-13-6-4-5-12(3)9-13/h4-11H,1-3H3,(H,17,19)(H2,18,20,21)
- InChIKey
- NGBFQHCMQULJNZ-UHFFFAOYSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


