| General Information of Drug (ID:
DR1884) |
| Drug Name |
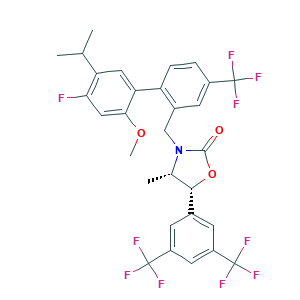
MK-0859
|
| Synonyms |
Anacetrapib; Anacetrapib (MK-0859); MK-0859; MK0859; P7T269PR6S; (4S,5R)-5-[3,5-Bis(trifluoromethyl)phenyl]-3-[[4'-fluoro-5'-isopropyl-2'-methoxy-4-(trifluoromethyl)biphenyl-2-yl]methyl]-4-methyl-1,3-oxazolidin-2-one; (4s,5r)-5-[3,5-bis(trifluoromethyl)phenyl]-3-({2-[4-fluoro-2-methoxy-5-(propan-2-yl)phenyl]-5-(trifluoromethyl)phenyl}methyl)-4-methyl-1,3-oxazolidin-2-one; 875446-37-0; CHEMBL1800807; MFCD16294903; UNII-P7T269PR6S
|
| Indication |
Atherosclerosis
[ICD11: BA80]
|
Phase 3
|
[1]
|
| Structure |
|
 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
637.5 |
Topological Polar Surface Area |
38.8 |
| Heavy Atom Count |
44 |
Rotatable Bond Count |
6 |
| Hydrogen Bond Donor Count |
0 |
Hydrogen Bond Acceptor Count |
13 |
| Cross-matching ID |
- PubChem CID
- 11556427
- PubChem SID
-
16658560
; 23677163
; 42698323
; 57376836
; 77084272
; 96025538
; 125240981
; 125299337
; 126671698
; 126731193
; 134338754
; 134449322
; 135252031
; 135723595
; 135727855
; 136340336
; 136354914
; 136367491
; 136368100
; 136988957
; 137140669
; 140430199
; 144115606
; 152258394
; 152344255
; 160647231
; 162011426
; 162038221
; 162205119
; 164023422
; 164194107
; 164835813
; 164835814
; 170499297
; 174007279
; 174531057
; 198963790
; 198991968
; 203355967
; 205994811
; 223381545
; 223435184
; 223683902
; 223704791
; 224000756
; 226842677
; 242591118
; 242786119
; 249736586
; 249821974
- CAS Number
-
- TTD Drug ID
- D07HLS
- Formula
- C30H25F10NO3
- Canonical SMILES
- CC1C(OC(=O)N1CC2=C(C=CC(=C2)C(F)(F)F)C3=CC(=C(C=C3OC)F)C(C)C)C4=CC(=CC(=C4)C(F)(F)F)C(F)(F)F
- InChI
- 1S/C30H25F10NO3/c1-14(2)22-11-23(25(43-4)12-24(22)31)21-6-5-18(28(32,33)34)9-17(21)13-41-15(3)26(44-27(41)42)16-7-19(29(35,36)37)10-20(8-16)30(38,39)40/h5-12,14-15,26H,13H2,1-4H3/t15-,26-/m0/s1
- InChIKey
- MZZLGJHLQGUVPN-HAWMADMCSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


