| General Information of Drug (ID:
DR0928) |
| Drug Name |
Letermovir
|
| Synonyms |
Letermovir; Letermovir [USAN:INN]; Prevymis; Prevymis (TN); (S)-{8-fluoro-2-[4-(3-methoxyphenyl)piperazine-1-yl]-3-(2-methoxy-5-trifluoromethylphenyl)-3,4-dihydroquinazoline-4-yl}acetic acid; 1H09Y5WO1F; 2-((4S)-8-Fluoro-2-(4-(3-methoxyphenyl)piperazin-1-yl)-3-(2-methoxy-5-(trifluoromethyl)phenyl)-4H-quinazolin-4-yl)acetic acid; 2-[(4S)-8-fluoro-2-[4-(3-methoxyphenyl)piperazin-1-yl]-3-[2-methoxy-5-(trifluoromethyl)phenyl]-4H-quinazolin-4-yl]acetic acid; 917389-32-3; AIC 246; AIC-246; AIC246; UNII-1H09Y5WO1F
|
| Indication |
Cytomegalovirus infection
[ICD11: 1D82]
|
Approved
|
[1]
|
| Structure |
|
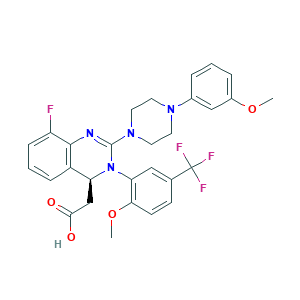 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
572.5 |
Topological Polar Surface Area |
77.8 |
| Heavy Atom Count |
41 |
Rotatable Bond Count |
7 |
| Hydrogen Bond Donor Count |
1 |
Hydrogen Bond Acceptor Count |
10 |
| Cross-matching ID |
- PubChem CID
- 45138674
- PubChem SID
-
92388531
; 103045327
; 120177785
; 123055444
; 123086332
; 137837293
; 163642903
; 175268695
; 210275200
; 210280838
; 224305963
; 226710579
; 243917611
- CAS Number
-
- TTD Drug ID
- D0G8PA
- Formula
- C29H28F4N4O4
- Canonical SMILES
- COC1=C(C=C(C=C1)C(F)(F)F)N2C(C3=C(C(=CC=C3)F)N=C2N4CCN(CC4)C5=CC(=CC=C5)OC)CC(=O)O
- InChI
- 1S/C29H28F4N4O4/c1-40-20-6-3-5-19(16-20)35-11-13-36(14-12-35)28-34-27-21(7-4-8-22(27)30)23(17-26(38)39)37(28)24-15-18(29(31,32)33)9-10-25(24)41-2/h3-10,15-16,23H,11-14,17H2,1-2H3,(H,38,39)/t23-/m0/s1
- InChIKey
- FWYSMLBETOMXAG-QHCPKHFHSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


