| General Information of Drug (ID:
DR1724) |
| Drug Name |
Zalcitabine
|
| Synonyms |
Zalcitibine; Cytidine, 2',3'-dideoxy-; Dideoxycytidine; Ro 24-2027/000; Ro-24-2027/000; WREGKURFCTUGRC-POYBYMJQSA-N; zalcitabine; 1-(2,3-Dideoxy-beta-D-ribofuranosyl)cytosine; 2',3'-DIDEOXYCYTIDINE; 2,3-dideoxycytidine; 4-Amino-1-((2R,5S)-5-(hydroxymethyl)tetrahydrofuran-2-yl)pyrimidin-2(1H)-one; 4-amino-1-[(2R,5S)-5-(hydroxymethyl)tetrahydrofuran-2-yl]pyrimidin-2(1H)-one; 6L3XT8CB3I; 7481-89-2; BRN 0654956; C9H13N3O3; CCRIS 692; CHEBI:10101; CHEMBL853; HIVID; HSDB 7156; MFCD00012188; NSC 606170; UNII-6L3XT8CB3I; ddC; ddCyd
|
| Indication |
Human immunodeficiency virus infection
[ICD11: 1C60]
|
Approved
|
[1]
|
| Structure |
|
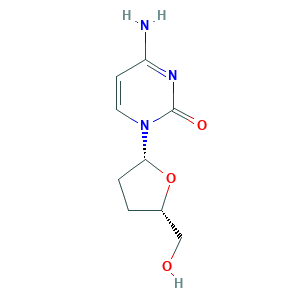 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
211.22 |
Topological Polar Surface Area |
88.2 |
| Heavy Atom Count |
15 |
Rotatable Bond Count |
2 |
| Hydrogen Bond Donor Count |
2 |
Hydrogen Bond Acceptor Count |
3 |
| Cross-matching ID |
- PubChem CID
- 24066
- PubChem SID
-
9416
; 595891
; 596441
; 596607
; 596679
; 601537
; 602107
; 878108
; 3259589
; 7847478
; 7980904
; 8167576
; 11467065
; 11468185
; 11486840
; 11504751
; 11528334
; 11532889
; 14797711
; 15171927
; 17389522
; 17404923
; 22391526
; 22431601
; 24278377
; 24862834
; 25819962
; 26719633
; 26752752
; 26752753
; 29291163
; 46386814
; 46488030
; 46507879
; 47216920
; 47216921
; 47589142
; 48185139
; 48334652
; 48416706
; 48422195
; 48422429
; 48423540
; 48424478
; 49681781
; 49693297
; 49699414
; 49734166
; 50105404
; 50105405
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D0Z9QR
- Formula
- C9H13N3O3
- Canonical SMILES
- C1CC(OC1CO)N2C=CC(=NC2=O)N
- InChI
- 1S/C9H13N3O3/c10-7-3-4-12(9(14)11-7)8-2-1-6(5-13)15-8/h3-4,6,8,13H,1-2,5H2,(H2,10,11,14)/t6-,8+/m0/s1
- InChIKey
- WREGKURFCTUGRC-POYBYMJQSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


