Details of the Drug Metabolized by Drug-Metabolizing Enzyme (DME)
| General Information of Drug (ID: DR2268) | ||||||
|---|---|---|---|---|---|---|
| Drug Name |
BRN-1907611
|
|||||
| Synonyms |
Alphanon; Bornan-2-one; Bornane, 2-oxo-; Camphor, (1R,4R)-(+)-; Camphor, synthetic; D-(+)-Camphor; DL-Bornan-2-one; DL-Camphor; Formosa camphor; Gum camphor; Kampfer; Laurel camphor; Matricaria camphor; Norcamphor, 1,7,7-trimethyl-; Root bark oil; Root bark spirit; Spirit of camphor; camphor; l-(-)-Camphor; (+)-Camphor; (+/-)-Camphor; 1,7,7-Trimethylbicyclo[2.2.1]heptan-2-one; 1,7,7-Trimethylnorcamphor; 2-Bornanone; 2-Camphanone; 2-Camphonone; 2-Keto-1,7,7-trimethylnorcamphane; 21368-68-3; 76-22-2; Bicyclo[2.2.1]heptan-2-one, 1,7,7-tri
|
|||||
| Indication | Inflammatory bowel disease [ICD11: DD72] | Phase 3 | [1] | |||
| Structure |
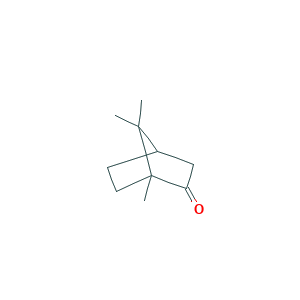 |
|||||
| 3D MOL | 2D MOL | |||||
| Pharmaceutical Properties | Molecular Weight | 152.23 | Topological Polar Surface Area | 17.1 | ||
| Heavy Atom Count | 11 | Rotatable Bond Count | 0 | |||
| Hydrogen Bond Donor Count | 0 | Hydrogen Bond Acceptor Count | 1 | |||
| Cross-matching ID |
|
|||||
| The Metabolic Roadmap of This Drug | |||||
|---|---|---|---|---|---|
| The Full List of Drug Metabolites (DM) of This Drug | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||
| The Full List of Metabolic Reaction (MR) of This Drug | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||
| Drug-Metabolizing Enzyme(s) (DME) Metabolizing This Drug | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.

