| General Information of Drug (ID:
DR1601) |
| Drug Name |
Tipranavir
|
| Synonyms |
Tipranavir; U-140690; Aptivus; PNU 140690; PNU-140690; ZZT404XD09; 174484-41-4; 3'-((1R)-1-((6R)-5,6-Dihydro-4-hydroxy-2-oxo-6-phenethyl-6-propyl-2H-pyran-3-yl)propyl)-5-(trifluoromethyl)-2-pyridinesulfonanilide; CHEBI:63628; N-(3-{(1R)-1-[(6R)-4-HYDROXY-2-OXO-6-PHENETHYL-6-PROPYL-5,6-DIHYDRO-2H-PYRAN-3-YL]PROPYL}PHENYL)-5-(TRIFLUOROMETHYL)-2-PYRIDINESULFONAMIDE; N-[3-[(1R)-1-[(2R)-6-hydroxy-4-oxo-2-phenethyl-2-propyl-3H-pyran-5-yl]propyl]phenyl]-5-(trifluoromethyl)pyridine-2-sulfonamide; TPV; UNII-ZZT404XD09
|
| Indication |
Human immunodeficiency virus infection
[ICD11: 1C60]
|
Approved
|
[1]
|
| Structure |
|
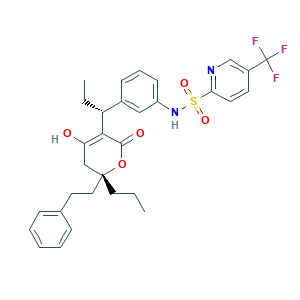 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
602.7 |
Topological Polar Surface Area |
114 |
| Heavy Atom Count |
42 |
Rotatable Bond Count |
11 |
| Hydrogen Bond Donor Count |
2 |
Hydrogen Bond Acceptor Count |
10 |
| Cross-matching ID |
- PubChem CID
- 54682461
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D0EV6T
- Formula
- C31H33F3N2O5S
- Canonical SMILES
- CCCC1(CC(=C(C(=O)O1)C(CC)C2=CC(=CC=C2)NS(=O)(=O)C3=NC=C(C=C3)C(F)(F)F)O)CCC4=CC=CC=C4
- InChI
- 1S/C31H33F3N2O5S/c1-3-16-30(17-15-21-9-6-5-7-10-21)19-26(37)28(29(38)41-30)25(4-2)22-11-8-12-24(18-22)36-42(39,40)27-14-13-23(20-35-27)31(32,33)34/h5-14,18,20,25,36-37H,3-4,15-17,19H2,1-2H3/t25-,30-/m1/s1
- InChIKey
- SUJUHGSWHZTSEU-FYBSXPHGSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


