Details of the Drug Metabolized by Drug-Metabolizing Enzyme (DME)
| General Information of Drug (ID: DR3115) | ||||||
|---|---|---|---|---|---|---|
| Drug Name |
LMP400
|
|||||
| Synonyms |
INDOTECAN; 915303-09-2; UNII-BTA69L5M8D; NSC-724998; BTA69L5M8D; 2,3-dimethoxy-6-(3-morpholinopropyl)-5H-[1,3]dioxolo[4',5':5,6]indeno[1,2-c]isoquinoline-5,12(6H)-dione; LMP-400; NSC724998; Indotecan(LMP400); Indotecan (LMP400); CHEMBL216462; SCHEMBL7712572; DTXSID90238613; BCP34018; EX-A3020; Indotecan hydrochloride (LMP-400); ZB1545; SB16793; HY-18351; CS-0007476; LMP400; LMP-400; LMP 400; Q27274872; 3-[(Morpholine)-1-propyl]-5,3-dimethoxy-8,9-meth ylenedioxy-5,11-dioxo-11H-indeno[1,2-c]isoquinoline
|
|||||
| Indication | Lymphoma [ICD11: 2A80-2A86] | Phase 1 | [1] | |||
| Neoplasm [ICD11: ICD11: 2A00-2F9Z] | Phase 1 | [2] | ||||
| Structure |
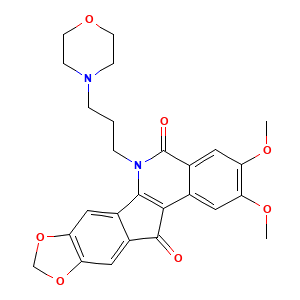 |
|||||
| 3D MOL | 2D MOL | |||||
| Pharmaceutical Properties | Molecular Weight | 478.5 | Topological Polar Surface Area | 86.8 | ||
| Heavy Atom Count | 35 | Rotatable Bond Count | 6 | |||
| Hydrogen Bond Donor Count | 0 | Hydrogen Bond Acceptor Count | 8 | |||
| Cross-matching ID |
|
|||||
| The Metabolic Roadmap of This Drug | |||||
|---|---|---|---|---|---|
| The Full List of Drug Metabolites (DM) of This Drug | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| The Full List of Metabolic Reaction (MR) of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.

