| General Information of Drug (ID:
DR1016) |
| Drug Name |
Mefloquine hydrochloride
|
| Synonyms |
Mefloquin; Mefloquina; Mefloquina [INN-Spanish]; Mefloquine [USAN:INN:BAN]; Mefloquinum; Mefloquinum [INN-Latin]; Mephloquine; Loriam; Mefloquin Hydrochloride; Mefloquine (hydrochloride); Mefloquine HCL; Mefloquine hydrochloride; Mephaquin; Ro 21-5998/001; WR 142490 hydrochloride; (AS)-rel-a-(2R)-2-Piperidinyl-2,8-bis(trifluoromethyl)-4-quinolinemethanol monohydrochloride; 326VC85GV6; 51773-92-3; DL-erythro-alpha-2-Piperidyl-2,8-bis(trifluoromethyl)-4-quinolinemethanol monohydrochloride; DSSTox_CID_27796; DSSTox_GSID_47819; DSSTox_RID_82568; EINECS 257-412-0; MLS001332559; NSC 157387; UNII-326VC85GV6; RTI1189-1-1; Racemic mefloquine; Ro 21-5998; Ro 215998; WR 142490; mefloquine; (-)-(11S,2'R)-erythro-Mefloquine; (-)-Mefloquine; (DL-erythro-alpha-2-Piperidyl-2,8-bis(trifluoromethyl)-4-quinolinemethanol; (S)-[2,8-bis(trifluoromethyl)quinolin-4-yl][(2R)-piperidin-2-yl]methanol; 53230-10-7; CHEBI:63687; CHEMBL172; HSDB 6853
|
| Indication |
Malaria
[ICD11: 1F40]
|
Approved
|
[1]
|
| Structure |
|
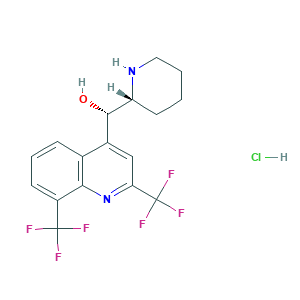 |
|
3D MOL is unavailable
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
414.8 |
Topological Polar Surface Area |
45.2 |
| Heavy Atom Count |
27 |
Rotatable Bond Count |
2 |
| Hydrogen Bond Donor Count |
3 |
Hydrogen Bond Acceptor Count |
9 |
| Cross-matching ID |
- PubChem CID
- 65329
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D0GQ7K
- Formula
- C17H17ClF6N2O
- Canonical SMILES
- C1CCNC(C1)C(C2=CC(=NC3=C2C=CC=C3C(F)(F)F)C(F)(F)F)O.Cl
- InChI
- 1S/C17H16F6N2O.ClH/c18-16(19,20)11-5-3-4-9-10(15(26)12-6-1-2-7-24-12)8-13(17(21,22)23)25-14(9)11;/h3-5,8,12,15,24,26H,1-2,6-7H2;1H/t12-,15+;/m1./s1
- InChIKey
- WESWYMRNZNDGBX-YLCXCWDSSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


