Details of the Drug Metabolized by Drug-Metabolizing Enzyme (DME)
| General Information of Drug (ID: DR1299) | ||||||
|---|---|---|---|---|---|---|
| Drug Name |
Pirfenidone
|
|||||
| Synonyms |
Pirespa; Pirfenidona; Pirfenidona [INN-Spanish]; Pirfenidone [USAN:INN]; Pirfenidonum; Pirfenidonum [INN-Latin]; 2(1H)-Pyridinone, 5-methyl-1-phenyl-; 5-Methyl-1-phenyl-1H-pyridin-2-one; 5-Methyl-1-phenyl-2(1H)-pyridone; 5-Methyl-1-phenyl-2-(1H)-pyridone; AMR 69; AMR-69; D7NLD2JX7U; Deskar; Esbriet; ISWRGOKTTBVCFA-UHFFFAOYSA-N; PIRFENIDONE; 5-methyl-1-phenyl-2(1H)-pyridinone; 5-methyl-1-phenyl-2-pyridinone; 5-methyl-1-phenylpyridin-2(1H)-one; 5-methyl-1-phenylpyridin-2-one; 53179-13-8; BRN 1526549; CHEBI:32016; UNII-D7NLD2JX7U
|
|||||
| Indication | Acute kidney failure [ICD11: GB60] | Approved | [1] | |||
| Structure |
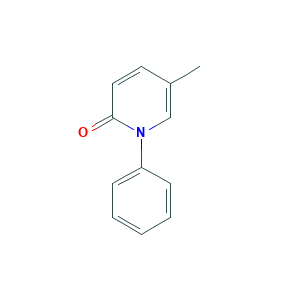 |
|||||
| 3D MOL | 2D MOL | |||||
| Pharmaceutical Properties | Molecular Weight | 185.22 | Topological Polar Surface Area | 20.3 | ||
| Heavy Atom Count | 14 | Rotatable Bond Count | 1 | |||
| Hydrogen Bond Donor Count | 0 | Hydrogen Bond Acceptor Count | 1 | |||
| Cross-matching ID |
|
|||||
| The Metabolic Roadmap of This Drug | |||||
|---|---|---|---|---|---|
| The Full List of Drug Metabolites (DM) of This Drug | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||
| The Full List of Metabolic Reaction (MR) of This Drug | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||
| Drug-Metabolizing Enzyme(s) (DME) Metabolizing This Drug | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.

