| General Information of Drug (ID:
DR1327) |
| Drug Name |
Pravastatin
|
| Synonyms |
Pravachol; Pravastatin [INN:BAN]; Pravastatin acid; Pravastatina; Pravastatina [Spanish]; Pravastatine; Pravastatine [French]; Pravastatinum; Pravastatinum [Latin]; Vasten; Eptastatin; KXO2KT9N0G; PRAVASTATIN SODIUM; pravastatin; (3R,5R)-3,5-dihydroxy-7-[(1S,2S,6S,8S,8aR)-6-hydroxy-2-methyl-8-{[(2S)-2-methylbutanoyl]oxy}-1,2,6,7,8,8a-hexahydronaphthalen-1-yl]heptanoic acid; 81093-37-0; CCRIS 7557; CHEBI:63618; CHEMBL1144; UNII-KXO2KT9N0G
|
| Indication |
Dyslipidaemia
[ICD11: 5C81]
|
Approved
|
[1]
|
| Structure |
|
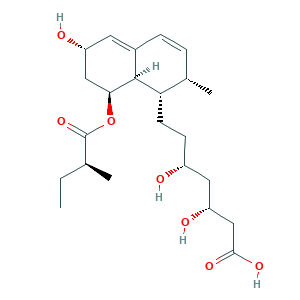 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
424.5 |
Topological Polar Surface Area |
124 |
| Heavy Atom Count |
30 |
Rotatable Bond Count |
11 |
| Hydrogen Bond Donor Count |
4 |
Hydrogen Bond Acceptor Count |
7 |
| Cross-matching ID |
- PubChem CID
- 54687
- PubChem SID
-
4962
; 7980374
; 8183732
; 10852022
; 14758460
; 14880512
; 34718661
; 46504851
; 48416457
; 49688762
; 49995722
; 50718915
; 57288813
; 57313644
; 85789083
; 85856288
; 93166931
; 96025096
; 102979949
; 104234199
; 104305598
; 117544687
; 118048679
; 124899367
; 126665388
; 129317935
; 134337841
; 135014686
; 135610132
; 135650848
; 137002685
; 142742106
; 152034648
; 152164597
; 160963523
; 164814679
; 165235255
; 174007284
; 175266850
; 175426985
; 175612187
; 179148899
; 184545482
; 187072512
; 210279284
; 210281607
; 223554816
; 223682192
; 223745588
; 226393956
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D02RQU
- Formula
- C23H36O7
- Canonical SMILES
- CCC(C)C(=O)OC1CC(C=C2C1C(C(C=C2)C)CCC(CC(CC(=O)O)O)O)O
- InChI
- 1S/C23H36O7/c1-4-13(2)23(29)30-20-11-17(25)9-15-6-5-14(3)19(22(15)20)8-7-16(24)10-18(26)12-21(27)28/h5-6,9,13-14,16-20,22,24-26H,4,7-8,10-12H2,1-3H3,(H,27,28)/t13-,14-,16+,17+,18+,19-,20-,22-/m0/s1
- InChIKey
- TUZYXOIXSAXUGO-PZAWKZKUSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


