| General Information of Drug (ID:
DR0948) |
| Drug Name |
Levothyroxine sodium
|
| Synonyms |
Levoroxine; Levothyroxine sodique; Levothyroxinum natricum; Novothyrox; Oroxine; Ro-thyroxine; Roxstan; Sodium L-thyroxine; Sodium levothyroxine; Sodium thyroxin; Sodium thyroxinate; Tetroid; Thyradin; Thyradin S; Thyro-Tabs; Thyronamin; Thyroxevan; Thyroxin sodium; Thyroxine sodium; Tiroidina; Tirosint; Tyrosint; Unithroid; l-Thyroxine monosodium salt; Dathroid; Eferox; Euthyrox; L-Thyroxine sodium; L-Thyroxine sodium salt; Laevoxin; Letter; Levaxin; Levo-T; Levolet; [125I]-(-)-Thyroxine; [125I]-CCRIS 6739; [125I]-HSDB 3108;80945-46-6; AC1LD8X6; DTXSID30349358; GTPL4627; [125I]-L-Thyroxin; [125I]-L-Thyroxine; [125I]-Laevothyroxinum (acid); [125I]-Levothyroxine; [125I]-Levothyroxinum (acid); [125I]-Synthroid; [125I]-T4; [125I]-T4 (Hormone); [125I]-THX; [125I]-Tetraiodothyronine; [125I]-Thryroxine, l-; [125I]-Thyratabs; [125I]-Thyrax; [125I]-Thyreoideum; [125I]-Thyroxin; [125I]-Thyroxinal; [125I]-Thyroxine; [125I]-Thyroxine (VAN); [125I]-Thyroxine (l); [125I]-Thyroxine iodine; [125I]thyroxine
|
| Indication |
Hypothyroidism
[ICD11: 5A00]
|
Approved
|
[1]
|
| Structure |
|
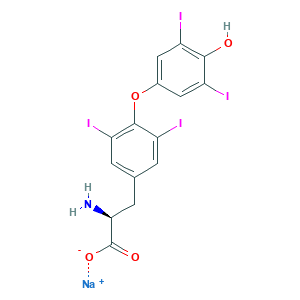 |
|
3D MOL is unavailable
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
798.85 |
Topological Polar Surface Area |
95.6 |
| Heavy Atom Count |
25 |
Rotatable Bond Count |
5 |
| Hydrogen Bond Donor Count |
2 |
Hydrogen Bond Acceptor Count |
5 |
| Cross-matching ID |
- PubChem CID
- 23666112
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D06RGG
- Formula
- C15H10I4NNaO4
- Canonical SMILES
- C1=C(C=C(C(=C1I)OC2=CC(=C(C(=C2)I)O)I)I)CC(C(=O)[O-])N.[Na+]
- InChI
- 1S/C15H11I4NO4.Na/c16-8-4-7(5-9(17)13(8)21)24-14-10(18)1-6(2-11(14)19)3-12(20)15(22)23;/h1-2,4-5,12,21H,3,20H2,(H,22,23);/q;+1/p-1/t12-;/m0./s1
- InChIKey
- YDTFRJLNMPSCFM-YDALLXLXSA-M
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


