Details of the Drug Metabolized by Drug-Metabolizing Enzyme (DME)
| General Information of Drug (ID: DR1939) | ||||||
|---|---|---|---|---|---|---|
| Drug Name |
Chrysin
|
|||||
| Synonyms |
Chrysin, 99+%; Chrysine; Crysin; FLAVONE, 5,7-DIHYDROXY-; RTIXKCRFFJGDFG-UHFFFAOYSA-N; chrysin; 3CN01F5ZJ5; 480-40-0; 4H-1-Benzopyran-4-one, 5,7-dihydroxy-2-phenyl-; 5,7-Dihydroxy-2-phenyl-4H-1-benzopyran-4-one; 5,7-Dihydroxy-2-phenyl-4H-benzo(b)pyran-4-one; 5,7-Dihydroxy-2-phenyl-4H-chromen-4-one; 5,7-Dihydroxy-2-phenyl-chromen-4-one; 5,7-Dihydroxyflavone; 5,7-dihydroxy-2-phenylchromen-4-one; BRN 0233276; CAS-480-40-0; CHEBI:75095; CHEMBL117; EINECS 207-549-7; MFCD00006834; NSC 407436; NSC-407436; NSC407436; UNII-3CN01F5ZJ5
|
|||||
| Indication | Discovery agent | Investigative | [1] | |||
| Structure |
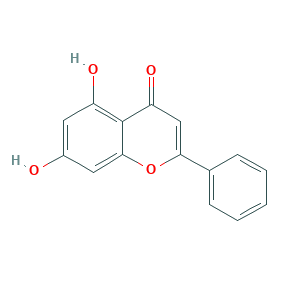 |
|||||
| 3D MOL | 2D MOL | |||||
| Pharmaceutical Properties | Molecular Weight | 254.24 | Topological Polar Surface Area | 66.8 | ||
| Heavy Atom Count | 19 | Rotatable Bond Count | 1 | |||
| Hydrogen Bond Donor Count | 2 | Hydrogen Bond Acceptor Count | 4 | |||
| Cross-matching ID | ||||||
| The Metabolic Roadmap of This Drug | |||||
|---|---|---|---|---|---|
| The Full List of Drug Metabolites (DM) of This Drug | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||
| The Full List of Metabolic Reaction (MR) of This Drug | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||
| Drug-Metabolizing Enzyme(s) (DME) Metabolizing This Drug | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Enzyme Kinetic Data of This Drug | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.

