| Synonyms |
Dihydronaringenin; Phloretol; S5J5OE47MK; phloretin; 1-Propanone, 3-(4-hydroxyphenyl)-1-(2,4,6-trihydroxyphenyl)-; 2',4',6'-Trihydroxy-3-(4-Hydroxyphenyl)propiophenone; 2',4',6'-Trihydroxy-3-(p-hydroxyphenyl)propiophenone; 3-(4-Hydroxyphenyl)-1-(2,4,6-trihydroxyphenyl)-1-propanone; 3-(4-Hydroxyphenyl)-1-(2,4,6-trihydroxyphenyl)propan-1-one; 60-82-2; CCRIS 7459; EINECS 200-488-7; MFCD00002288; NSC 407292; NSC407292; UNII-S5J5OE47MK; beta-(p-Hydroxyphenyl)phloropropiophenone
|
| Cross-matching ID |
- PubChem CID
- 4788
- PubChem SID
-
4036
; 477076
; 841166
; 885022
; 3152290
; 6900044
; 7979953
; 8141738
; 8144209
; 8152940
; 11111666
; 11111667
; 11341604
; 11361787
; 11364189
; 11366751
; 11369313
; 11372170
; 11374941
; 11377475
; 11484537
; 11487189
; 11488497
; 11490851
; 11492986
; 11495109
; 11537656
; 14750659
; 17405570
; 22425581
; 24278652
; 24771297
; 24887442
; 26613299
; 26679936
; 26725279
; 26747651
; 26753632
; 26759355
; 29204475
; 29223872
; 46500461
; 47440053
; 47736274
; 47736275
; 47885227
; 47959549
; 48334288
; 48421899
; 49760075
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D0HD2G
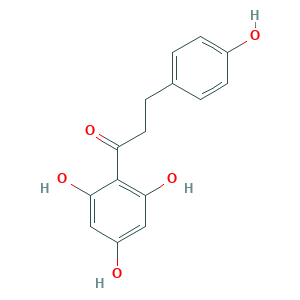
- Formula
- C15H14O5
- Canonical SMILES
- C1=CC(=CC=C1CCC(=O)C2=C(C=C(C=C2O)O)O)O
- InChI
- 1S/C15H14O5/c16-10-4-1-9(2-5-10)3-6-12(18)15-13(19)7-11(17)8-14(15)20/h1-2,4-5,7-8,16-17,19-20H,3,6H2
- InChIKey
- VGEREEWJJVICBM-UHFFFAOYSA-N
|