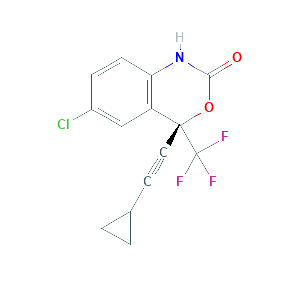
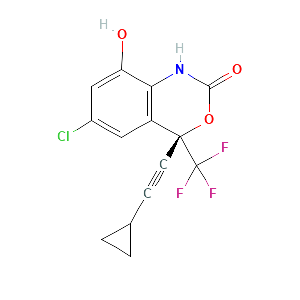
| References |
| 1 |
Clinical pharmacokinetics and drug-drug interactions of Elbasvir/Grazoprevir. Eur J Drug Metab Pharmacokinet. 2018 Oct;43(5):509-531.
|
| 2 |
The Activity of Members of the UDP-Glucuronosyltransferase Subfamilies UGT1A and UGT2B is Impaired in Patients with Liver Cirrhosis. Clin Pharmacokinet. 2023 Jun 16. doi: 10.1007/s40262-023-01261-3.
|
| 3 |
Induction of CYP3A4 by efavirenz in primary human hepatocytes: comparison with rifampin and phenobarbital. J Clin Pharmacol. 2004 Nov;44(11):1273-81.
|
| 4 |
Synthetic and natural compounds that interact with human cytochrome P450 1A2 and implications in drug development. Curr Med Chem. 2009;16(31):4066-218.
|
| 5 |
Evaluation of CYP2B6 induction and prediction of clinical drug-drug interactions: considerations from the IQ consortium induction working group-an industry perspective. Drug Metab Dispos. 2016 Oct;44(10):1720-30.
|