Details of the Drug Metabolized by Drug-Metabolizing Enzyme (DME)
| General Information of Drug (ID: DR1893) | ||||||
|---|---|---|---|---|---|---|
| Drug Name |
LAU-7b
|
|||||
| Synonyms |
FENRETINIDE; Fenretinida; Fenretinida [Spanish]; Fenretinide [USAN:INN]; Fenretinidum; Fenretinidum [Latin]; McN-R-1967; N-(4-Hydroxyphenyl)retinamide; Retinamide, N-(4-hydroxyphenyl)-; Retinoic acid p-hydroxyanilide; all-trans-4'-Hydroxyretinanilide; 15-[(4-hydroxyphenyl)amino]retinal; 4-(hydroxyphenyl)retinamide; 4-HPR; 4-Hydroxyphenyl retinamide; 4-hydroxy(phenyl)retinamide; 4-hydroxyphenylretinamide; 4HPR; 65646-68-6; CCRIS 3260; N-(4-Hydroxyphenyl)all-Trans Retinamide; Retinoic acid p-hydroxyphenylamide; UNII-187EJ7QEXL
|
|||||
| Indication | Macular degeneration [ICD11: 9B78] | Phase 3 | [1] | |||
| Structure |
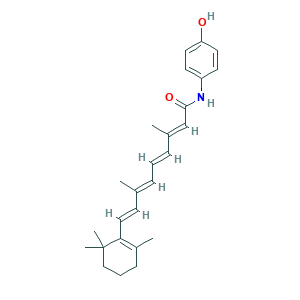 |
|||||
| 3D MOL | 2D MOL | |||||
| Pharmaceutical Properties | Molecular Weight | 391.5 | Topological Polar Surface Area | 49.3 | ||
| Heavy Atom Count | 29 | Rotatable Bond Count | 6 | |||
| Hydrogen Bond Donor Count | 2 | Hydrogen Bond Acceptor Count | 2 | |||
| Cross-matching ID |
|
|||||
| The Metabolic Roadmap of This Drug | |||||
|---|---|---|---|---|---|
| The Full List of Drug Metabolites (DM) of This Drug | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| The Full List of Metabolic Reaction (MR) of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drug-Metabolizing Enzyme(s) (DME) Metabolizing This Drug | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.

