Details of the Drug Metabolized by Drug-Metabolizing Enzyme (DME)
| General Information of Drug (ID: DR1515) | ||||||
|---|---|---|---|---|---|---|
| Drug Name |
Sulfinpyrazone
|
|||||
| Synonyms |
Sulfinpirazona [INN-Spanish]; Sulfinpyrazine; Sulfinpyrazon; Sulfinpyrazonum; Sulfinpyrazonum [INN-Latin]; Sulfoxyphenylpyrazolidine; Sulphinpyrazone; USAF GE-13; Anturan; Anturane; Anturane (TN); Anturanil; Anturano; Anturen; Anturidin; Apo-Sulfinpyrazone; Diphenylpyrazone; Enturan; Enturen; sulfinpyrazone; (+/-)-SULFINPYRAZONE; 1,2-Diphenyl-4-(2-(phenylsulfinyl)ethyl)pyrazolidine-3,5-dione; 4-(Phenylsulfoxyethyl)-1,2-diphenyl-3,5-pyrazolidinedione; 57-96-5; G 28315; NSC 75925
|
|||||
| Indication | Uncontrolled gout [ICD11: FA25] | Approved | [1] | |||
| Structure |
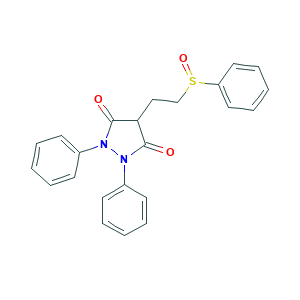 |
|||||
| 3D MOL | 2D MOL | |||||
| Pharmaceutical Properties | Molecular Weight | 404.5 | Topological Polar Surface Area | 76.9 | ||
| Heavy Atom Count | 29 | Rotatable Bond Count | 6 | |||
| Hydrogen Bond Donor Count | 0 | Hydrogen Bond Acceptor Count | 4 | |||
| Cross-matching ID |
|
|||||
| The Metabolic Roadmap of This Drug | |||||
|---|---|---|---|---|---|
| The Full List of Drug Metabolites (DM) of This Drug | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| The Full List of Metabolic Reaction (MR) of This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drug-Metabolizing Enzyme(s) (DME) Metabolizing This Drug | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.

