Details of the Drug Metabolized by Drug-Metabolizing Enzyme (DME)
| General Information of Drug (ID: DR0929) | ||||||
|---|---|---|---|---|---|---|
| Drug Name |
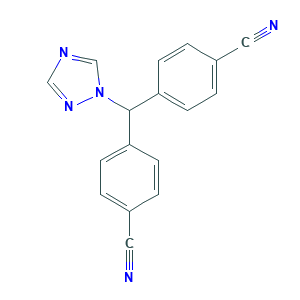
Letrozole
|
|||||
| Synonyms |
Letrozol; Letrozole; Femara; Femera; CGS 20267; CGS-20267; UNII-7LKK855W8I; LETRAZOLE; 112809-51-5; 4,4'-((1h-1,2,4-triazol-1-yl)methylene)dibenzonitrile; 4,4'-(1H-1,2,4-Triazol-1-ylmethylene)dibenzonitrile; 4,4'-(1H-1,2,4-triazol-1-ylmethanediyl)dibenzonitrile; 4,4'-(1h-1,2,4-triazol-1-ylmethylene)bisbenzonitrile; 4-[(4-CYANOPHENYL)(1H-1,2,4-TRIAZOL-1-YL)METHYL]BENZONITRILE; 7LKK855W8I; Benzonitrile, 4,4'-(1H-1,2,4-triazol-1-ylmethylene)bis-; C17H11N5; CHEBI:6413; CHEMBL1444; HSDB 7461
|
|||||
| Indication | Breast cancer [ICD11: 2C60] | Approved | [1] | |||
| Structure |
 |
|||||
| 3D MOL | 2D MOL | |||||
| Pharmaceutical Properties | Molecular Weight | 285.3 | Topological Polar Surface Area | 78.3 | ||
| Heavy Atom Count | 22 | Rotatable Bond Count | 3 | |||
| Hydrogen Bond Donor Count | 0 | Hydrogen Bond Acceptor Count | 4 | |||
| Cross-matching ID |
|
|||||
| The Metabolic Roadmap of This Drug | |||||
|---|---|---|---|---|---|
| The Full List of Drug Metabolites (DM) of This Drug | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||
| The Full List of Metabolic Reaction (MR) of This Drug | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||
| Drug-Metabolizing Enzyme(s) (DME) Metabolizing This Drug | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.

