| General Information of Drug (ID:
DR0557) |
| Drug Name |
Ebastine
|
| Prodrug Info |
Ebastine is the prodrug of Carebastine
|
| Synonyms |
Ebastel; Ebastin; Ebastina [Spanish]; Ebastinum [Latin]; Estivan; Evastel; Kestin; Kestine; LAS W-090; RP 64305; Bactil; RP-64305; TQD7Q784P1; ebastine; 1-(4-tert-butylphenyl)-4-[4-(diphenylmethoxy)piperidin-1-yl]butan-1-one; 1-[4-(1,1-Dimethylethyl)phenyl]-4-[4-(diphenylmethoxy)-1-piperidinyl]-1-butanone; 4'-tert-Butyl-4-(4-(diphenylmethoxy)piperidino)butyrophenone; 4-Diphenylmethoxy-1-(3-(4-tert-butylbenzoyl)propyl)piperidine; 90729-43-4; C32H39NO2; CHEMBL305660; MFCD00865661; NCGC00164603-01; UNII-TQD7Q784P1
|
| Indication |
Irritable bowel syndrome
[ICD11: DD91]
|
Phase 4
|
[1]
|
| Structure |
|
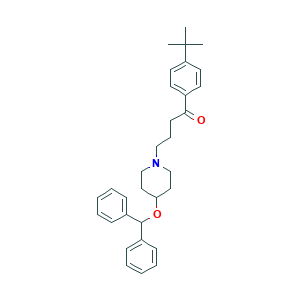 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
469.7 |
Topological Polar Surface Area |
29.5 |
| Heavy Atom Count |
35 |
Rotatable Bond Count |
10 |
| Hydrogen Bond Donor Count |
0 |
Hydrogen Bond Acceptor Count |
3 |
| Cross-matching ID |
- PubChem CID
- 3191
- ChEBI ID
-
- CAS Number
-
- Formula
- C32H39NO2
- Canonical SMILES
- CC(C)(C)C1=CC=C(C=C1)C(=O)CCCN2CCC(CC2)OC(C3=CC=CC=C3)C4=CC=CC=C4
- InChI
- 1S/C32H39NO2/c1-32(2,3)28-18-16-25(17-19-28)30(34)15-10-22-33-23-20-29(21-24-33)35-31(26-11-6-4-7-12-26)27-13-8-5-9-14-27/h4-9,11-14,16-19,29,31H,10,15,20-24H2,1-3H3
- InChIKey
- MJJALKDDGIKVBE-UHFFFAOYSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


