| General Information of Drug (ID:
DR0671) |
| Drug Name |
Etravirine
|
| Synonyms |
Etravirine; Etravirine (TMC125); Intelence; R-165335; R165335; TMC 125; TMC-125; TMC125; 0C50HW4FO1; 269055-15-4; 4-((6-Amino-5-bromo-2-((4-cyanophenyl)amino)pyrimidin-4-yl)oxy)-3,5-dimethylbenzonitrile; 4-({6-AMINO-5-BROMO-2-[(4-CYANOPHENYL)AMINO]PYRIMIDIN-4-YL}OXY)-3,5-DIMETHYLBENZONITRILE; 4-[[6-AMINO-5-BROMO-2-[(4-CYANOPHENYL)AMINO]-4-PYRIMIDINYL]OXY]-3,5-DIMETHYLBENZONITRILE; C20H15BrN6O; CHEBI:63589; CHEMBL308954; UNII-0C50HW4FO1
|
| Indication |
Human immunodeficiency virus infection
[ICD11: 1C60]
|
Approved
|
[1]
|
| Structure |
|
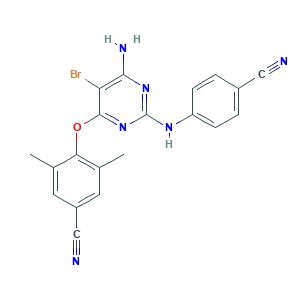 |
|
3D MOL
|
2D MOL
|
| Pharmaceutical Properties |
Molecular Weight |
435.3 |
Topological Polar Surface Area |
121 |
| Heavy Atom Count |
28 |
Rotatable Bond Count |
4 |
| Hydrogen Bond Donor Count |
2 |
Hydrogen Bond Acceptor Count |
7 |
| Cross-matching ID |
- PubChem CID
- 193962
- PubChem SID
-
642445
; 832707
; 7885445
; 10261828
; 12015430
; 14856802
; 33514683
; 47206062
; 50755335
; 57397726
; 58108352
; 92765305
; 92765307
; 93375510
; 93619697
; 99443662
; 103268452
; 103999716
; 113457302
; 118844113
; 125346599
; 126617154
; 126632876
; 126665857
; 131308179
; 134224563
; 134338670
; 135171617
; 135611140
; 135693135
; 136946574
; 136980023
; 137006543
; 139263775
; 143497676
; 144115317
; 152035774
; 152234808
; 152258935
; 160647779
; 160658773
; 160968275
; 162009794
; 162011438
; 162200353
; 162849709
; 163894003
; 164831834
; 170483270
; 170503235
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D0BP9C
- Formula
- C20H15BrN6O
- Canonical SMILES
- CC1=CC(=CC(=C1OC2=NC(=NC(=C2Br)N)NC3=CC=C(C=C3)C#N)C)C#N
- InChI
- 1S/C20H15BrN6O/c1-11-7-14(10-23)8-12(2)17(11)28-19-16(21)18(24)26-20(27-19)25-15-5-3-13(9-22)4-6-15/h3-8H,1-2H3,(H3,24,25,26,27)
- InChIKey
- PYGWGZALEOIKDF-UHFFFAOYSA-N
|
|
|
|
|
|
|
|
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.


