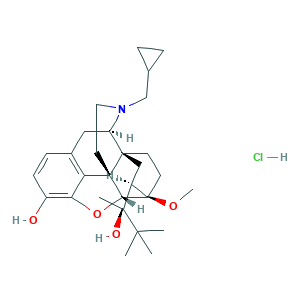
| Synonyms |
Belbuca; Buprenex (TN); Buprenorphine; Buprenorphine Depot; Buprex; CHEBI:652822; CL 112,302; CL 112302; CL-112302; EINECS 258-396-8; Finibron; MR 56; NIH 8805; NIH-8805; Norspan; RX 6029-M; RX 6029-M HCl; RX-6029-M; Transtec; UM 952; UM-952; UNII-56W8MW3EN1; Buprenex; Buprenophine; Buprenorfina; Buprenorfina [INN-Spanish]; Buprenorphinum; Buprenorphinum [INN-Latin]; CAM2038; CHEBI:3216; EINECS 257-950-6; Probuphenine; Probuphine; RX 6029M; Temgesic (TN); UNII-40D3SCR4GZ; (-)-buprenorphine; 21-cyclopropyl-7alpha-[(S)-1-hydroxy-1,2,2-trimethylpropyl]-6,14-endo-ethano-6,7,8,14-tetrahydrooripavine; 40D3SCR4GZ; 6029-M; Buprel; 21-Cyclopropyl-7alpha-((S)-1-hydroxy-1,2,2-trimethylpropyl)-6,14-endo-ethano-6,7,8,14-tetrahydrooripavine hydrochloride; 53152-21-9; 56W8MW3EN1; BEMA LA; BEMA-buprenorphine; BUPRENORPHINE HYDROCHLORIDE
|
| Cross-matching ID |
- PubChem CID
- 3033050
- ChEBI ID
-
- CAS Number
-
- TTD Drug ID
- D06AWE
- Formula
- C29H42ClNO4
- Canonical SMILES
- CC(C)(C)C(C)(C1CC23CCC1(C4C25CCN(C3CC6=C5C(=C(C=C6)O)O4)CC7CC7)OC)O.Cl
- InChI
- 1S/C29H41NO4.ClH/c1-25(2,3)26(4,32)20-15-27-10-11-29(20,33-5)24-28(27)12-13-30(16-17-6-7-17)21(27)14-18-8-9-19(31)23(34-24)22(18)28;/h8-9,17,20-21,24,31-32H,6-7,10-16H2,1-5H3;1H/t20-,21-,24-,26+,27-,28+,29-;/m1./s1
- InChIKey
- UAIXRPCCYXNJMQ-RZIPZOSSSA-N
|